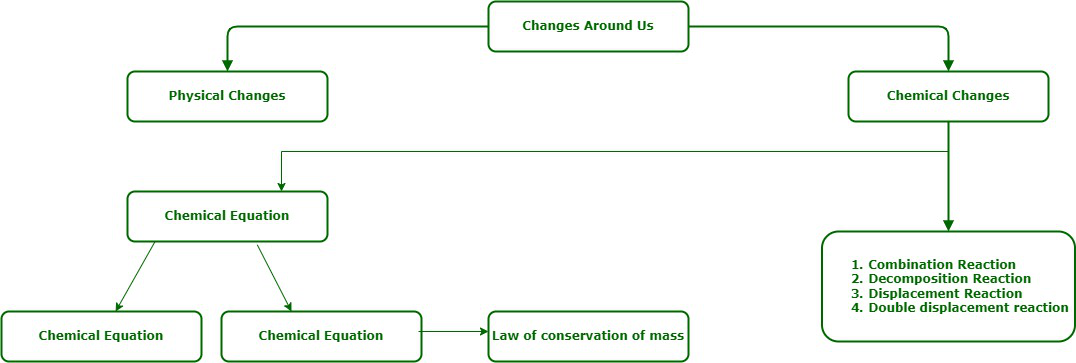
编写和平衡化学方程式
化学反应是反应物原子内的键断裂,产物原子内的取代基键结晶,从而结晶出取代基的过程。借助参与化学反应的物质的符号和分子式来表示化学反应的方法称为化学反应。化学反应与化学变化有关。当发生化学变化时,会发生化学反应。化学反应通常是不可逆的,会形成新的产物。原子在化学反应中重新排列。
- The substance which combines or reacts is known as a reactant.
- The new substance produced in a reaction is called a product.
A chemical equation is a concise method of representing a chemical reaction.
化学反应的特点是,
- 气体的演变
- 沉淀物的形成
- 颜色变化
- 温度变化
- 状态变化
平衡和不平衡的化学方程式
- 在平衡的化学方程式中,反应物和产物具有相同数量的不同元素的原子。
- 在不平衡的化学方程式中,反应物和产物中一种或多种元素的原子数不相等。
- 在平衡的化学方程式中,反应物和产物具有相同质量的不同元素。
- 在不平衡方程中,反应物和产物具有不同质量的不同元素。
- 反应物中不同类型的原子数应等于产物中相同类型的原子数。
- 平衡化学方程式以满足化学反应中的质量守恒定律。
Never change the formula of an element or compound to balance an equation.
使方程两边不同类型原子的数量相等的过程称为方程平衡。
提供有关化学反应的更多信息的方程式称为信息量更大或信息量更大的方程式。化学反应可以通过三种方式获得更多信息:
- 通过指示反应物和产物的“物理状态”。
- 通过指示反应中发生的“热变化”。
- 通过指明反应发生的“条件”。
写平衡化学方程式的重要例子
编写化学方程式的方程式必须遵循以下步骤:Step 1. Write the chemical reaction in the form of a word equation with the reactants on the left and the products on the right. Step 2. Substitute the symbols and formulas for all the reactants and products into the word equation. Step 3. Balance the equation by multiplying the symbols and formulas by the smallest possible number of digits. Step 4. However, make the equation extra informative by indicating temperature changes, if possible, denoting the physical states of the reactants and products. Nevertheless, this step can be avoided if you do not have sufficient information about the physical states, heat fluctuations, and reaction conditions.
记住这些事情,让我们解决一个例子:
示例:写出下列反应的平衡方程式: 甲烷在氧气中燃烧生成二氧化碳和水。
解决方案:
Step 1: Write the chemical reaction in the form of a word equation with the reactants on the left and the products on the right.
This reaction can be written as a one-term equation:
Carbon dioxide + Water → Methane + Oxygen
- Formula of methane is CH3
- Formula of oxygen is O₂
- Formula of carbon dioxide is CO₂
- Formula of water is H₂O
Step 2: Substitute the symbols and formulas for all the reactants and products into the word equation.
On writing the formulas of all the substances in the above word equation, we get:
CH4 + O2 → CO2 + H₂O
Let us calculate the structure of different atoms in the reactants and products: In reactants In products 1 1 4 2 2 3 Number of C atoms Number of H atoms Number of O atoms
Step 3: Balance the equation by multiplying the symbols and formulas by the smallest possible number of digits.
The composition of carbon atoms is the indistinguishable on both sides (1 each) but the composition of hydrogen tittles isn’t equal to that of the oxygen atoms of copper. There are 4 hydrogen atoms on the reactant side but only 2 hydrogen atoms on the product side. on the right side. To deposit 4 hydrogen atoms on the right, we multiply H₂O by 2 and write 2H2O. Therefore,
CH4 + 2O2 → CO2 + H₂O
Counting the number of various atoms on both the sides again, we get : In reactants In products Number of C atoms 1 1 Number of H atoms 4 4 Number of O atoms 2 4
Now, only the number of oxygen atoms is unequal. There are 2 oxygen atoms on the left but 4 on the right. To have 4 oxygen atoms on the left, we multiply O₂ by 2 and write 2O₂.
CH4 + 2O2 →CO2 + 2H₂O
Let’s count the number of different atoms on both sides once again In reactants In products Number of C atoms 1 1 Number of H atoms 4 4 Number of O atoms 4 4
In this chemical equation, there are equal number of different types of atoms in the reactants and products, so it is a balanced equation.
详细描述以更好地理解上述示例 -
通过指示反应物和产物的物理状态以及反应中发生的热量变化,可以使上述方程提供更多信息,如下所述:
- 甲烷是一种气体- CH 4 (g)
- 氧气是一种气体-O₂(g)
- 二氧化碳是一种气体 - CO₂ (g)
H₂O的物理状态如何?
如果反应发生在水性介质中,则 H2O 处于液态,我们将其记为 HO(1)。如果反应发生在气相中,则 H 2 O 处于气态并表示为 H 2 O (g)。在这种情况下,甲烷气体燃烧成氧气以形成二氧化碳气体和水蒸气或水流。所以这里的水是气态的,是H2O(g)。如果我们把所有反应物和产物的物理状态,那么方程可以写成 -
CH 4 (g) + 2O 2 (g) → CO 2 (g) +2H 2 O(g)
现在,在这个反应中发生的热量发生了变化。甲烷在氧气中燃烧生成二氧化碳和水时,也会产生大量热量,所以是放热反应。放热反应通过在产品一侧写上“+加热”标志来表示。所以,上面的等式最终可以写成:
CH 4 (g) + 2O 2 (g) → CO 2 (g) +2H 2 O(g) + 热
这个方程现在给出了这些反应物和产物的物理状态以及反应中发生的热量变化,因此它是一个信息量更大的方程。
示例问题
问题1:列出你周围的任何四个变化,通过这些变化我们可以确定是否发生了化学反应。
回答:
- Respiration of animals,
- Digestion of food,
- Burning of coal, and
- Rusting of iron.
问题 2:提及任何三个有助于确定化学反应是否发生的观察结果。写一个活动,至少可以证明列出的两个评论是正确的。
回答:
Observations:
- Change in state,
- Change in color,
- Evolution of a gas,
- Change in temperature.
Activity:
- Some zinc granules are taken in a conical flask.
- A small amount of dilute hydrochloric acid is poured over it.
- It is observed that the zinc granules start decreasing in size and hydrogen gas is released.
- The temperature of the flask also rises slightly.
问题 3:提及可以使一般化学方程式提供更多信息的三种方法。
回答:
There are three ways to make a general chemical equation more informative:
- By balancing the chemical reaction, the number of atoms of each reactant and product can be made equal.
- More information about the physical states of reactants and products can be obtained by referring to their physical states (s, l, g, aq).
- The heat must be specified in favor of the reactants or products in order to identify an endothermic or exothermic reaction.
问题4:Seema买了这包薯片,打开了。突然,他的朋友夏努来了。她开始玩,却忘了吃薯条。第二天吃薯条的时候,他觉得味道不好,身体不舒服。他让妈妈带他去看医生。医生告诉他,这是因为吃了不新鲜的薯片。他给了西玛几天内痊愈的药物。
(a) 为什么薯片在密封包装中保鲜时间更长?
(b) 为什么开包芯片坏了?
(丙)你从这段经文中学到什么价值?
回答:
(a) This is because sealed packets of chips are usually filled with a gas such as nitrogen, which prevents the chips from oxidizing.
(b) The fats and oils present in the open packet of chips get oxidized. They become rancid and their smell and taste change.
(c) Food items should be stored in airtight containers or in the refrigerator to prevent oxidation of the fats and oils present in them. Once the chips packets/food item gets spoiled, their smell and taste change. They should not be eaten because they are not good for our health.
问题 5:H₂O 在化学反应中的物理状态是什么?
回答:
If the reaction takes place in an aqueous medium, then H₂O is in the liquid state and we write H-O(1) for it. If the reaction takes place in the vapor phase, then H₂O is in the gaseous state and is denoted as H₂O (g).